
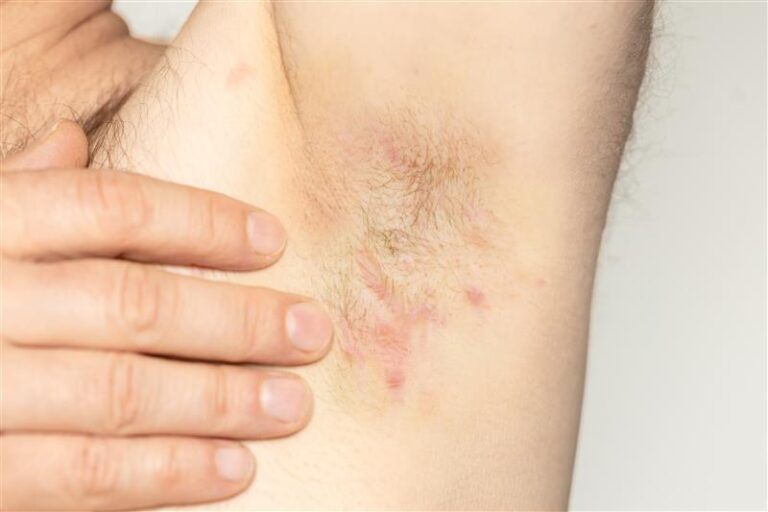
hidradenitis suppurativa (HS) can cause painful skin symptoms that come and go. Researchers want to better understand how to steady these waters. By joining the LATITUDE HS Study, you could help create ripples of change that reach far beyond your own experience. See if the study is right for you.

The LATITUDE HS Study is evaluating the effectiveness and safety of an investigational oral treatment for adults with HS.
Participants will receive the investigational oral treatment or a placebo (a substance that looks like the investigational treatment but does not contain any active ingredients) while completing various study tests and procedures. Participants originally assigned to the placebo group will have the opportunity to switch to the investigational oral treatment during the study. All study treatments will be given orally.
Neither participants nor the study staff will know if participants are receiving the investigational treatment or placebo. Participants should plan to attend up to 12 scheduled study visits over the course of approximately 61 weeks.

By signing up you agree to receive text messages and emails about this study. You can unsubscribe at any time. Text messages and data rates may apply. You also agree that StudyKIK may provide your personal information to the research site to help determine if you qualify for this study. Please refer to StudyKIK's Privacy Policy and Terms of Service for more information.
Keep in mind that participation is entirely voluntary. If you do decide to take part in a study, you may change your mind about participating at any time.
We use cookies to optimize site functionality and give you the best experience. Necessary cookies enable core functionality. The website cannot function properly without these cookies and can only be disabled by changing your browser preferences.
For more detailed information on the cookies we use, please check our Privacy Policy.
By continuing to access this website, you are giving us consent to collect cookies.
You are now leaving latitudehsstudy.com and being redirected to a website that is not operated by Clinical Trial Media Inc.